Human tissue samples
We enrolled 60 patients who have undergone radiotherapy and surgery at the First Affiliated Hospital of Nanjing Medical University, Nanjing China. The study was conducted in accordance with the Declaration of Helsinki, and approved by the Ethics Committee of the First Affiliated Hospital of Nanjing Medical University (Protocol code: 2023-SR-777). All enrolled patients provided written informed consent prior to participation, acknowledging their understanding of the study’s purpose, procedures, and potential risks. Tumor tissues collected from the participants were confirmed as lung cancer tissues by two pathologists.
Cell culture
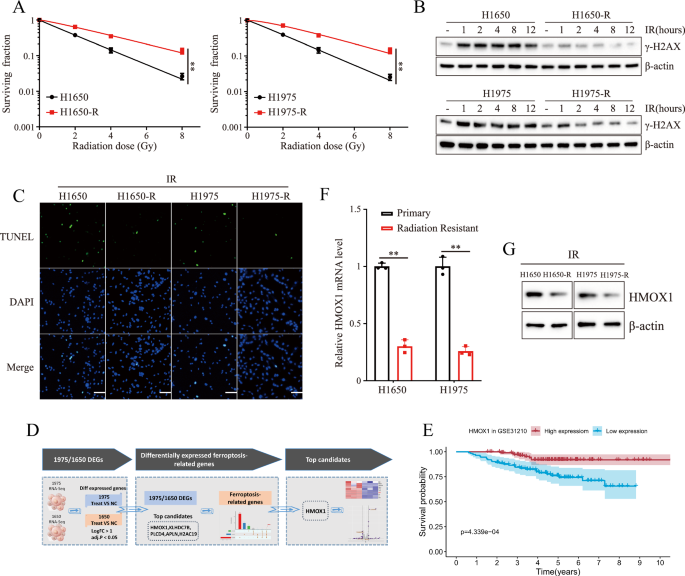
The human non-small cell lung cancer (NSCLC) cell lines H1650, H1975 and HEK293T cells were obtained from the Shanghai Cell Bank (Chinese Academy of Science, Shanghai, China).To establish radioresistant cell lines, H1650R and H1975R, H1650 and H1975 cells were subjected to 5 cycles of irradiation with 8 Gy, followed by two additional cycles of irradiation at a dose rate of 1 Gy/min, with doses of 8 Gy for the first 5 cycles and 10 Gy for the last 2 cycles (8, 8, 8, 8, 8, 10, 10 Gy) using X-rays. Cells were maintained in Dulbecco’s Modified Eagle Medium (DMEM) supplemented with 10% fetal bovine serum (FBS) and 1% penicillin-streptomycin antibiotics (Gibco Laboratories; Termo Fisher Scientific, Massachusetts, USA) at 37 °C in a humidified atmosphere of 5% CO2 and 95% air (Galaxy 170 R, Eppendorf, Hamburg, Germany). All cell lines were authenticated using Short Tandem Repeat (STR) profiling and confirmed to be free of mycoplasma contamination.
Cell viability and colony formation assay
For cell viability assays, cells were seeded in a 96-well plate at a density of 1 × 104 cells per well. Cell viability was assessed using the Cell Counting Kit-8 (CCK-8) assay (Beyotime, C0037, Shanghai, China) according to the manufacturer’s instructions. After incubation for 2 h at 37 °C, the plate was analyzed using a microplate reader, and absorbance of the wells was measured at 450 nm. To determine cell survival fraction, cells were seeded at a density of 1 × 106 cells per dish and cultured in for two weeks. Subsequently, cells were washed with phosphate-buffered saline (PBS), fixed with 4% formaldehyde for 10 minutes, and stained with a 0.5% crystal violet solution for 30 minutes. Colonies consisting of more than 50 stained cells were counted as surviving colonies.
Xenograft tumor mouse model
The experiments on animals were approved by the Institutional Animal Care and Use Committee of Nanjing Medical University under IACUC approval No. 2501032. Nude mice (6 weeks old, male, BALB/C-nu/nu) obtained from the Nanjing Medical University Animal Center were selected randomly and used for ectopic tumor development. To establish a subcutaneous tumor model, H1650R cells transfected with a lentiviral vector carrying the human HMOX1 gene (lvHMOX1) (H1650R-lvHMOX1) or its negative control (H1650R-lvNC)(1 × 107 cells in 100 μL) were injected into the left flank of nude mice. Additionally, H1650R-shHMOX1/H1650R-shNC cells were also injected at the same site. All mice were divided into 8 groups based on the type of inoculated cells and radiation treatment (IR). When the tumor size reached approximately 50 mm3, the tumor sites received 10 Gy X-rays irradiation for 3 consecutive days, while the rest of the mouse body was shielded with a lead plate. Once the tumor volume reached approximately 1000 mm3, the mice were euthanized via cervical dislocation. Xenograft tumors were then excised, photographed, and divided into two parts. One half was fixed and embedded in paraffin for subsequent immunofluorescence staining, while the other half was rapidly frozen in liquid nitrogen for future preservation. All animal experiments were approved by the Animal Experimental Ethical Inspection Committee of the First Affiliated Hospital, Nanjing Medical University.
To establish pulmonary metastasis models, NSG mice were intravenously injected with 1 × 106 H1650 GL cells suspended in 100 μL of phosphate-buffered saline (PBS) on day 0. One week post inoculation of tumor cells, bioluminescence imaging (BLI) was performed on the mice. The mice were categorized into 8 groups based on the combination of cell type and IR treatment. For pulmonary metastases in NSG mice, a standard radiotherapy regimen was administered with a dose of 2 Gy per treatment on day 5, delivered every other day for a total of five treatments. On day 7, Mice were monitored by using BLI.
RNA Isolation and sequencing analysis
Total RNA was extracted from cells using Trizol reagent (Invitrogen, San Diego, CA, USA) according to the manufacturer’s protocol. RNA sequencing (RNA-seq) analysis was performed using single-stranded circular DNA derived from mRNA. Single-stranded circular DNA molecules are replicated via rolling circle amplification, generating DNA nanoballs (DNBs) containing multiple copies of the DNA. Sufficient quality DNBs were then loaded onto patterned nanoarrays using high-intensity DNA nanochip technique and sequenced using combinatorial Probe-Anchor Synthesis (cPAS). Differentially expressed genes (DEGs) were identified by comparing RNA-seq data from H1650-R/H1650 and H1975-R/H1975 cells. Transcripts with a p-value < 0.05 were classified as differentially expressed mRNAs (DEmRNAs).
Biochemical assay and Lipid ROS imaging
Intracellular Fe2+ ion levels were measured using a Ferro-orange kit (Dojingo, Molecular Technologies Inc., Shanghai, China) according to the manufacturer’s instruction. In addition, the concentrations or activity of MDA in the specified samples were measured using commercially available enzyme-linked immunosorbent assay (ELISA) kits (Abcam, ab118970), following the manufacturer’s instructions. Notably, cells were irradiated with 4 Gy of X-rays and subsequently incubated in serum-free medium for 4 hours prior to these assays.
For Lipid ROS imaging, Cells were seeded onto 6-well chamber slides at a density of 5 × 105 cells per well and incubated for 24 hours. After washing with PBS, the slides were incubated with a solution containing 2 mM BODIPY 581/591 C11 (D3861, Invitrogen) and 200 nM Mito Tracker Deep Red FM (Invitrogen) for 20 minutes. Subsequently, the slides were visualized using a confocal microscope (Carl Zeiss Microscopy GmbH, Germany).
Protein half-life assay
Cells transfected with the specified plasmids were subjected to treatment with the protein synthesis inhibitor CHX (100 μg/ml; Sigma-Aldrich) for the designated time periods prior to sample collection.
Western Blot
Total protein of cells were extracted using RIPA lysate buffer (Beyotime Biotechnology, China) according to the manufacturer’s instruction. Protein samples (20 μg) were separated by SDS-PAGE and transferred to PVDF membranes. Subsequently, the membranes were blocked with 5% BSA in TBST solution for one hour at room temperature. followed by incubation with the primary antibody at 4 °C overnight. After three 10-min washes with TBST, the membranes were incubated with secondary antibodies diluted in the blocking buffer for one hour at room temperature. Following three additional washes with TBST, enhanced chemiluminescence (ECL) was performed using an ECL kit (Beyotime Biotechnology, Shanghai, China). Densitometric analysis of each band was conducted using ImageJ software to quantify the results. The following primary antibodies were used in this study: anti-γ-H2AX (catalog ab81299), anti-HMOX1 (catalog ab189491), anti-ACSL4 (catalog ab155282), anti-GPX4 (catalog ab125066), anti-FTH1 (catalog ab183781), anti-β-actin (catalog ab8226), anti-Histone (catalog ab1791), anti-NRF2 (catalog ab62352) and anti-KEAP1 (catalog ab119403) from Abcam; anti-Flag (catalog 14793), anti-HA (catalog 3724) and anti–Myc (catalog 2276) from CST.
Real-time quantitative PCR
RNA extraction from cells was performed using the RNA Kit I (Omega, Norcross, GA, USA). Following the manufacturer’s instructions, reverse transcription of the isolated total RNA into cDNA was conducted using a 20 μl reaction volume of the qRT-PCR Kit (Tiangen, Beijing, China). For HMOX1 gene, the forward primer was 5’-CGCTGAGGATCCATGGACTACAAAGACGAT-3’ and the reverse primer was 5’-CGTGCATCTAGATCACATGGCATAAAGCCC-3’. For β-Actin gene, the forward primer was 5’-CATGTACGTTGCTATCCAGGC-3’ and the reverse primer was 5’-CTCCTTAATGTCACGCACGAT-3’.
Immunofluorescence (IF) and immunohistochemistry (IHC) assay
For IF assay, cells were fixed with 4% formaldehyde, permeabilized with 0.25% Triton X-100, and blocked with 1% BSA for one hour at room temperature. After blocking, samples were incubated with the designated primary antibodies overnight at 4 °C. Following a PBS rinse, cells were incubated with secondary antibodies for two hours at 37 °C. Subsequently, cells were treated with DAPI (Beyotime Biotechnology, Haimen, China) for 10 min. The secondary antibodies used were goat anti-mouse IgG FITC, goat anti-rabbit IgG Cy3, and goat anti-mouse IgG Cy5. Fluorescence images of at least three randomly selected fields were captured using a fluorescence microscope (Nikon Eclipse CI-S, Tokyo, Japan).
For the IHC assay, subcutaneous tumor tissues were deparaffinized and rehydrated by passing through a series of decreasing alcohol concentrations. Antigen retrieval was performed using sodium citrate buffer. Tumor sections were blocked with a solution containing 1% BSA, 0.25% Triton X-100, and 3% H2O2 in PBS for 1 h at room temperature. Subsequently, sections were incubated with the designated primary antibodies overnight at 4 °C, followed by counterstaining with hematoxylin.
Co-immunoprecipitation (Co-IP) assay
Whole cell lysates (WCL) were prepared according to the manufacturer’s instructions (Beyotime Biotechnology) and centrifuged at 10,000 × g for 10 min at 4 °C. Subsequently, 1 ml of the supernatant was incubated with 1 μg of anti-USP7 antibody, anti-KEAP1 antibody, and anti-IgG antibody for 16 hours. Next, 20 μl of fresh protein A/G plus agarose beads (Santa Cruz Biotechnology, Shanghai, China) were added and the mixture was incubated overnight at 4 °C. The samples were then spun down, washed four times with immunoprecipitation buffer, and subjected to SDS-PAGE for fractionation followed by Western blot analysis.
Plasmid construction and transfection
All shRNAs, Flag-tagged USP7, Flag-tagged USP7(C223A), Myc-tagged USP7, Flag-tagged KEAP1 plasmids were purchased from Vigene Biosciences (Shandong, China). These plasmids were constructed by inserting the respective cDNA sequences into the pCMV-MCS expression vector at the AsisI and M1uI restriction sites using cloning techniques. Additionally, HA-ubiquitin plasmid and its mutant (K48R, K63R) plasmid were obtained from Hanyin Biotechnology (Shanghai, China) by cloning the corresponding full-length human DNA sequence into the HA-pcDNA 3.1(+) vector. Lentiviral vectors (Ubi-CMV-HMOX1-SV40-puro) were purchased from Genechem (Shanghai, China). The empty vector served as the negative control. For all transfection experiments, Lipofectamine 3000 reagents (L3000015, Invitrogen, Eugene, OR, USA) were used according to the manufacturer’s protocols.
Mass spectrometry
Liquid chromatography (LC) coupled with tandem mass spectrometry (MS) analysis was conducted by BGI Tech Solutions Co., Ltd (BGI Shenzhen, Guangdong, China). Protein samples were subjected to trypsin digestion at a protein-to-enzyme ratio of 1:20 and incubated at 37 °C for 4 h. For each sample, an amount equivalent to 2–5 mg of protein was loaded onto the LC–MS/MS system.
GST pull-down assays
The bacterial expression plasmid pGEX-4T-1 containing GST-KEAP1, GST-USP7-WT, and GST-USP7-C223A was used to express the proteins in Escherichia coli BL21. Protein expression was induced by adding 0.4 mM isopropyl β-d-1-thiogalactopyranoside at 16 °C. The purified proteins were obtained using GST beads (Sigma-Aldrich) following the manufacturer’s instructions. Glutathione-Sepharose 4B beads (GE Healthcare) were then used to immobilize bacterial-expressed GST, GST-KEAP1, GST-USP7-WT, or GST-USP7-C223A. These beads were incubated with Flag-USP7 WT or Flag-USP7 C223A proteins expressed in HEK293T cells for 2 hours at 4 °C. Following incubation, the complexes were washed with GST-binding buffer at least four times, eluted by boiling in SDS-PAGE loading buffer, and subsequently analyzed by immunoblotting using the specified antibodies.
Statistical analyses
Statistical analyses were performed using Prism 8.0.2 software (GraphPad Software, USA). Student’s t-test was used to compare quantitative data between two groups, while one-way analysis of variance (ANOVA) was employed for multiple group comparisons. The subcutaneous tumor diameters in each group were analyzed using a two-way ANOVA. All results were expressed as mean ± standard deviation (SD) and replicated in at least three independent experiments. A p-value less than 0.05 was considered statistically significant.