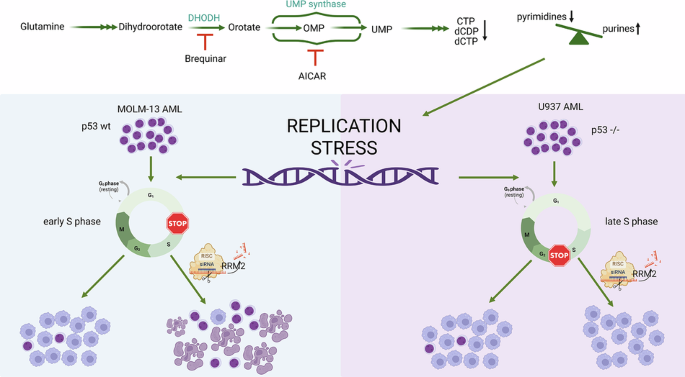
Differentiation therapy offers a promising approach in acute myeloid leukemia (AML) by overcoming the developmental block that maintains leukemic blasts. Increasing evidence indicates that DNA replication stress can promote differentiation rather than cytotoxicity; however, the metabolic mechanisms linking replication stress to differentiation remain poorly defined. Here, we investigated how perturbations in nucleotide metabolism regulate replication stress–driven differentiation. Using metabolomic and functional analyses in AML cell lines, we show that agents inducing differentiation through replication stress, including 5-aminoimidazole-4-carboxamide ribonucleoside (AICAr), dihydroorotate dehydrogenase (DHODH) inhibition, and low-dose cytarabine, converge on disruption of nucleotide pool balance. Low-dose AICAr induced a pyrimidine–purine imbalance, S phase arrest, and enhanced differentiation, whereas high-dose reduced these effects. Although brequinar and cytarabine altered nucleotide metabolism through distinct mechanisms, differentiation induced by all agents was abolished by supplementation with high levels of ribo- and deoxyribonucleosides, confirming that nucleotide imbalance is a central driver. We further identify ribonucleotide reductase (RNR) as a critical modulator of this process. Replication stress induced context-dependent regulation of RNR subunits, with RRM2 upregulated in p53-mutant U937 cells and the p53-responsive RRM2B isoform predominating in p53-wild-type MOLM-13 cells. Consistent with these differences, RRM2 depletion enhanced differentiation in U937 cells without affecting viability but impaired differentiation and survival in MOLM-13 cells. These findings position nucleotide metabolism as a key regulator of AML differentiation and suggest that combining RNR-targeted and checkpoint-modulating strategies could optimize therapeutic responses.
The alternative text for this image may have been generated using AI.