Cell lines and reagents
Human HNSCC cell lines SCC-15, SCC-25, and QLL-1, WS1 (human fibroblasts) were obtained from the American Type Culture Collection (ATCC) and cultured in Dulbecco’s modified Eagle’s medium (DMEM) supplemented with 10% fetal bovine serum (FBS), 1% penicillin-streptomycin, and 1% glutamine. The cells were maintained at 37 °C in a humidified incubator with 5% CO₂.
5-Fluorouracil (5-FU; Cat. F6627), Cisplatin (Cat. P4394), and Puromycin (Cat. P9620) were purchased from Sigma-Aldrich (St. Louis, MO, USA). U0126 (ERK inhibitor; Cat. No. S1102) and WP1066 (STAT3 inhibitor; Cat. No. HY-15312) were purchased from MedChemExpress (MCE, USA).
Lentiviral transduction and HERC1 knockdown
Stable knockdown of HERC1 was achieved using a short hairpin RNA (shRNA) construct targeting HERC1 (sh.HERC1; sequence: CAGATTGTTGAGCGCTTATTT; GACTGCTTTATGACCATATTA; GAGCGCAAGCCATGATCTATA; Vector Builder, USA) with scrambled shRNA as a control (sh.Scr). Transfection was performed using Lipofectamine™ 3000 reagent (Cat. L3000015, Invitrogen) following the manufacturer’s protocol. After transfection, the cells were selected using puromycin (5 μg/mL; Sigma-Aldrich) for 7 days. Knockdown efficiency was validated by Western blotting analysis.
Cell growth and proliferation
For cell counting, 1 × 103 cells transduced with sh.HERC1 were seeded in 6-well plates. Cells were harvested and counted by trypan blue exclusion at 24, 48, and 72 h. The growth was plotted over time.
For the proliferation assay, 5 × 103 cells/well were seeded in 96-well plates. After 24, 48, and 72 h, the WST-8 reagent (Cat. WQ3000, WST-8 Cell Viability Assay Kit) was added to each well following the manufacturer’s instructions. Absorbance was measured at 450 nm using a microplate reader and the data were analyzed accordingly.
Western blotting analysis
Total protein was extracted using RIPA buffer (sc-24948, Santa Cruz) containing protease and phosphatase inhibitors (Cat. 5872, Cell Signaling). Protein lysates (20 µg) were resolved by SDS-PAGE and transferred to nitrocellulose membranes. Membranes were incubated with primary antibodies against HERC1 (Cat. Sc-393950, Santa Cruz), CD44 (Cat. A1351, Abclonal; Cat. 3570, Cell Signaling), Nanog (Cat. A22625, Abclonal), Sox-2 (Cat. 23064, Cell Signaling), Oct4 (Cat. A7920, Abclonal), c-Myc (Cat. sc-42, Santa Cruz; Cat. AB32072, Abcam), Slug (Cat. 9585, Cell Signaling), Snail (Cat. 5243, Abclonal), Zeb1 (Cat. A5600, Abclonal), N-cadherin (Cat. 1MA1-159, Invitrogen), E-cadherin (Cat. AB15148, Abcam), Vimentin (Cat. MA3-745, Invitrogen; Cat. AB92547, Abcam), p-STAT3 (Y705) (Cat. sc-8059, Santa Cruz; Cat. 9145, Cell Signaling), STAT3 (Cat. ab68153, abcam), cleaved Caspase-3 (Cat. 9661, Cell Signaling), Bcl-2 (Cat. 13-8800, Invitrogen), p44/42 MAPK (Erk1/2) (Cat. 4695, Cell Signaling), ERK1 (sc-376852, Santa Cruz), Phospho-SAPK/JNK (Thr183/Tyr185) (Cat. 4668, Cell Signaling), JNK1 (sc-1648, Santa Cruz), Phospho-p38 MAPK (Thr180/Tyr182) (Cat. 4511, Cell Signaling), p38 MAPK (Cat. 9212, Cell Signaling), and β-actin (Cat. MAB8929, R&D system) overnight at 4 °C. After incubation with horseradish peroxidase-conjugated secondary antibodies, the bands were visualized using ECL substrate (Cat. LF-QC01060, AbFrontier).
Magnetic cell sorting (MACS)
CD44+ cells were isolated from the dissociated tumor spheroids using anti-CD44 MicroBeads (Cat. 130-095-194, Miltenyi Biotec) following the manufacturer’s instructions. Briefly, CD44+ cells (spheroids) were enzymatically dissociated into single-cell suspensions using Accutase (cat. A1110501; Life Technologies), and incubated with anti-CD44 magnetic beads at 4 °C for 15 min. The labeled cells were then passed through MS columns placed in a magnetic field (cat. 130-042-201, Miltenyi Biotec), and CD44⁺ cells were collected from the column. The purity of the isolated population was assessed by flow cytometry using a fluorophore-conjugated anti-CD44 antibody, and only samples with >90% CD44⁺ purity were used for downstream analysis.
Spheroid and organoid formation assays
For spheroid assays, 1 × 10⁴ cells were seeded in spheroid medium in low-attachment plates and cultured for up to 3–5 days with Advanced DMEM/F12, B27 (1X; Cat. 17504044, Gibco), N2 (1X), EGF (10 ng/mL; Cat. 17502048, Gibco), and bFGF (10 ng/mL; Cat. GF003AF, Sigma Aldrich) as spheroid culture conditions.
For organoid formation [32, 33], CD44⁺ cells were isolated by Magnetic-Activated Cell Sorting (MACS; Miltenyi Biotec) and overlaid with DMEM/F12 supplemented with penicillin/streptomycin, 10 mmol/L HEPES, GlutaMAX, 1X B27 (Cat. 17504044, Gibco), 1X N2 (Cat. 17502048, Gibco), 1.25 mmol/L N-acetylcysteine (Cat. A9165, Sigma-Aldrich), 0.05 mg/mL Epidermal Growth Factor (EGF; Cat. 9644, Sigma Aldrich), 0.1 mg/mL Fibroblast Growth Factor-basic (FGF-basic; Cat. GF003AF, Sigma Aldrich), 0.01 mmol/L gastrin I (Cat. G9145, Sigma Aldrich), 10 mmol/L Nicotinamide (Cat. N0636, Sigma Aldrich), 10 mmol/L Y-27632 (Cat. Y0503, Sigma Aldrich), and SB202190 (Cat. S7067, Sigma Aldrich); 1 mmol/L Prostaglandin E2 (Cat. 2296, Tocris Bioscience, Tocris Bioscience); 0.1 mg/mL Fibroblast Growth Factor-10 (FGF-10; Cat. 100-26, PeproTech); 0.5 mmol/L A83-01 (Cat. 2939, R&D Systems), and L-WRN Conditioned Media (Wnt3A, Rspondin 1, mNoggin; SCM105, Sigma) in 24-well plates. The organoid culture medium was completely refreshed every 3–4 days. Organoids were passaged every week at a 1:3–1:6 split ratio by removing them from Matrigel using BD Cell Recovery Solution (Cat. 354253, BD Biosciences) following the manufacturer’s instructions and transferring them to fresh Matrigel (Cat. 356255, BD Biosciences). Organoid numbers and sizes were quantified using the ImageJ software (NIH) [34].
Immunocytochemistry
WS1 fibroblasts and spheroids were fixed with 4% paraformaldehyde for 15 min, permeabilized with 0.1% Triton X-100 in PBS for 10 min. and blocked with 1% BSA in PBS for 1 h. Cells were incubated overnight at 4 °C with CD44 (FITC-conjugated; Cat. 130-113-334, Miltenyi Biotec), Sox-2 (Cat. 23064, cell Signaling), Slug (Cat. 9585, Cell Signaling), or FAP (Cat. sc-65398, Santa Cruz) antibodies, followed by Alexa Fluor 488/594/647–conjugated secondary antibodies (Cat. A-11008; Cat. A32744; Cat. A32728TR, Life Technologies). Nuclei were counterstained with DAPI (Cat. F6057, Sigma), and images were acquired using a Zeiss confocal microscope and processed with Imaris 7.6.
Migration and invasion assays
Cell migration and invasion were assessed using Transwell assays with Boyden chambers (8 µm pore size; Corning). For invasion assays, the upper surfaces of the inserts were coated with Matrigel (Cat. 354234), whereas the migration assays were performed without Matrigel. A total of 2 × 10⁴ cells were suspended in serum-free medium and seeded into the upper chamber. The lower chamber was filled with medium containing 10% FBS or fibroblasts as chemo-attractants. After 24–72 h of incubation at 37 °C, non-migrated or non-invaded cells were removed from the upper surface of the membrane using a cotton swab. Cells that had migrated or invaded the lower surface were fixed and stained using a staining kit (Cat. 898125, Diff Quick stain set) and counted in five randomly selected fields per insert under a light microscope.
Drug sensitivity and apoptosis assays
To assess chemotherapeutic sensitivity, CD44⁺ organoids were treated with increasing concentrations of 5-fluorouracil (5-FU; 0.01–100 µM; Cat. F6627, Sigma-Aldrich) or cisplatin (0.01–100 µM; Cat. P4394, Sigma-Aldrich) for 24–72 h. Drug treatments were performed in an organoid culture medium under standard conditions. Cell viability was measured using a WST-8 Cell Viability Assay Kit (Cat. WQ3000, UMUST R&D Co.) following the manufacturer’s instructions. Absorbance was recorded at 450 nm using a microplate reader, and dose-response curves were generated to calculate IC₅₀ values.
Apoptosis was assessed by IHC/IF staining of cleaved Caspase-3 in paraffin-embedded organoid sections, and western blotting analysis of cleaved Caspase-3 and Bcl-2 protein levels. For IHC and IF, organoids were fixed in 4% paraformaldehyde, embedded in paraffin, sectioned, and stained with an anti-cleaved Caspase-3 antibody (Cat. 9664, 1:200 dilution, Cell Signaling), and an anti-Bcl-2 antibody (Cat. 13-8800, Invitrogen). For Western blotting, total protein was extracted using RIPA buffer (sc-24948, Santa Cruz) supplemented with protease and phosphatase inhibitors (Cat. 5872, Cell Signaling) and analyzed by Western blotting.
Immunohistochemistry (IHC) and immunofluorescence (IF)
Formalin-fixed, paraffin-embedded xenograft and lung metastasis tissues were sectioned (3 μm), deparaffinized, rehydrated, and subjected to heat-induced antigen retrieval in citrate buffer (pH 6.0; Cat. 005000, Invitrogen). After blocking with 1% BSA, sections were incubated overnight at 4 °C with primary antibodies against HERC1 (Cat. PA5-62033, Thermo Scientific), phospho-STAT3 (Y705) (Cat. 9145, Cell Signaling), CD44 (Cat. 3570, Cell Signaling), Sox-2 (Cat. 14962, Cell Signaling), Ki-67 (Cat. 9449, Cell Signaling), cleaved Caspase-3 (Cat. 9661, Cell Signaling) or pan-Cytokeratin (Cat. 67306, Cell Signaling).
For IHC, HRP-conjugated secondary antibodies were applied, visualized with DAB (Cat. SK-4100, Vector Labs.), and counterstained with hematoxylin.
For IF, Alexa Fluor 488/594/647–conjugated secondary were used (Cat. A-11008; Cat. A32744; Cat. A32728TR, Life Technologies), nuclei were counterstained and mounted with DAPI (Cat. F6057, Sigma). Images were captured using a Zeiss confocal microscope under identical settings, and quantitative analysis was performed on ≥ 5 random high-power fields per sample using Imaris 7.6 or ImageJ.
Mouse xenograft and metastasis assays
All animal experiments were conducted in accordance with protocols approved by the Institutional Animal Care and Use Committee (IACUC) of Kangbuk Samsung Hospital (25-144-A1-N). For subcutaneous xenograft assays, 5 × 10⁶ sh.Scr or sh.HERC1 cells suspended in 100 μL of PBS:Matrigel (1:5) were injected into the flanks of 6-week-old NOD/SCID mice. Tumor growth was monitored every 3 days using calipers, and tumor volume was calculated using the following formula: (length × width²)/2.
For experimental metastasis assays, 1 × 10⁶ CD44⁺-derived organoids were resuspended in sterile PBS and injected into the lateral tail vein of NOD/SCID mice. After 3 weeks, the mice were euthanized and the lungs were harvested, fixed in 10% neutral-buffered formalin, embedded in paraffin, and sectioned for hematoxylin and eosin (H&E) staining, immunohistochemistry (IHC), and immunofluorescence (IF) to assess metastatic burden.
Co-culture system with fibroblasts
Human fibroblasts (WS-1) were cultured in DMEM supplemented with 10% FBS. For indirect co-culture experiments, 2 × 10⁴ fibroblasts per well were seeded into the lower chamber of a Transwell system (0.4 μm pore size), while CD44⁺ cells were seeded in the upper chamber. For three-dimensional (3D) direct co-culture, 2 × 10⁴ fibroblasts and 5 × 103 CD44⁺ organoids (4:1) were embedded together in Matrigel domes and cultured for 7 days. Organoid morphology and invasive behavior were assessed using bright-field microscopy.
Enzyme-linked immunosorbent assay (ELISA)
IL-6 concentration in conditioned media (CM) was measured using a human IL-6 Quantikine ELISA kit (R&D Systems, Cat. D6050), following the manufacturer’s protocol. CM was collected from fibroblasts, cancer cell lines, and CD44⁺ cells cultured individually or in co-culture systems (indirect Transwell or 3D Matrigel) for 48–72 h. Before collecting, CM was centrifuged at 300 × g for 5 min to remove cellular debris and stored at −80 °C until analysis. Absorbance was measured at 450 nm (with correction at 570 nm) using a microplate reader (SPARK, TECAN KOEA), and IL-6 concentrations were calculated using a standard curve generated with recombinant human IL-6 provided in the kit.
Cytokine treatment and blocking experiments
To modulate IL-6 signaling, CD44⁺ cells and organoids were treated with recombinant human IL-6 (20 ng/mL; R&D Systems, Cat. 206-IL) or an anti-IL-6 neutralizing antibody (1 µg/mL; Cat. MAB206, R&D Systems) for 72 h. Treatments were performed in a low-serum (0.5% FBS) culture medium to minimize background cytokine activity. For blocking experiments, cells were pre-incubated with low serum concentration for 2 h before IL-6 stimulation. After 72 h, cells and organoids were harvested for protein extraction or fixed for IF analysis. Western blotting and IF were used to evaluate the downstream signal activation and target protein expression, respectively.
Human tumor microarray (TMA) analysis
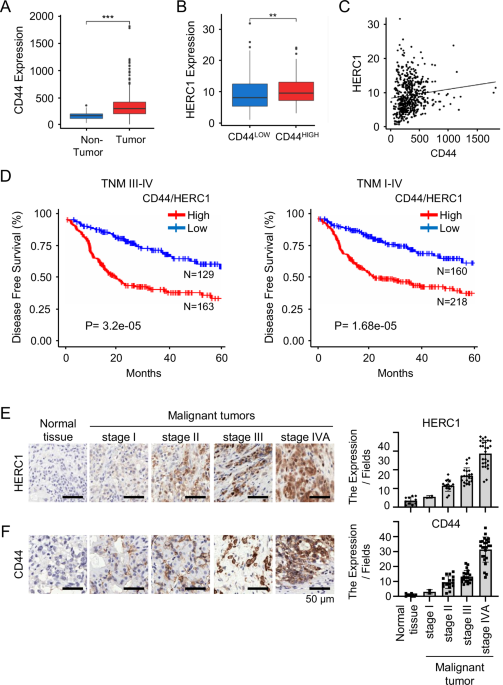
Commercial tissue microarray (TMA) slides (Cat. HN802d, TissueArray) containing HNSCC tissues at various stages and normal tissue controls were stained with antibodies against HERC1, CD44, and fibroblast activation protein (FAP). IHC was performed using standard protocols with diaminobenzidine as the chromogen. For each sample, five representative fields were scored for marker expression. Stained slides were digitally scanned using a whole-slide scanner (Pannoramic 150 Digital Scanner) at 20× and 40× magnifications to generate high-resolution images.
Image analysis was conducted using ImageJ software (NIH), allowing quantification of staining intensity, positive area, and cell counts. For IF staining, nuclei were counterstained with DAPI and used as a reference for cell segmentation and co-localization analysis. Quantitative data were derived from three independent experiments and presented as mean ± standard deviation (SD).
Data acquisition and processing
TCGA-HNSC bulk RNA-seq were downloaded using the R package TCGAbiolinks. Specifically, the GDCquery() and GDCdownload() functions were used to retrieve HTSeq-Counts from The Cancer Genome Atlas (TCGA) Head and Neck Squamous Cell Carcinoma (HNSC) cohort. Clinical metadata, including patient survival information, were retrieved from cBioPortal(http://www.cbioportal.org) for consistency and completeness.
Single-cell RNA-seq data from primary and metastatic HNSC tissues (GSE181919) were obtained from the Gene Expression Omnibus (GEO). The UMI count matrix and corresponding barcode-level metadata were loaded into R and preprocessed using the Seurat package (v4.3.0). Cell type annotation was based on the original metadata, and fibroblasts were subset for downstream analysis. For IL-6 expression analysis, fibroblasts from NL (normal adjacent tissue) and LP (leukoplakia) tissues were considered as normal fibroblasts, while those from CA (primary tumor) and LN (lymph node metastasis) tissues were classified as cancer-associated fibroblasts (CAFs).
Expression-based stratification
For CD44-based analysis, TCGA-HNSC patients were divided into high and low groups using the median expression level of CD44 as the cutoff. For combined CD44 and HERC1 analysis, the expression values of CD44 and HERC1 were first averaged within each patient, and the overall mean of these per-patient averages was used to define the cutoff. Patients with values above this mean were categorized as high, and those below as low.
Survival analysis
Overall survival analysis was performed using clinical metadata from the cBioPortal TCGA-HNSC cohort, which includes survival time (in months) and vital status (alive or deceased). Patients were grouped based on gene expression levels, using the same stratification strategy described above (combined CD44/HERC1 average). Kaplan–Meier survival curves were generated using the survival and survminer R packages. Samples with missing survival or expression data were excluded from the analysis.
Differential expression and gene set enrichment analysis
Differential gene expression analysis between high and low groups was performed using the limma-voom pipeline. Genes with a B-statistic greater than 5 were considered significantly differentially expressed and were used to construct ranked gene lists for gene set enrichment analysis (GSEA). GSEA was conducted using the clusterProfiler R package (v4.6.2) with Gene Ontology Biological Process (GO:BP) as the reference gene set collection. Genes were ranked by their B-statistic, and enriched terms with a false discovery rate (FDR) < 0.05 were considered statistically significant. Enrichment plots were visualized using gseaplot2() or custom ggplot2-based visualizations.
Statistical analysis
Data are presented as mean ± standard deviation (SD). All experiments were performed in triplicates. Statistical significance was determined using Student’s t-test or one-way ANOVA followed by Tukey’s post hoc test, or Wilcoxon rank-sum test for non-parametric comparisons. A P < 0.05 was considered statistically significant. GraphPad Prism 9, ImageJ, Microsoft Excel, and ggplot() in R were used for all the analyses.