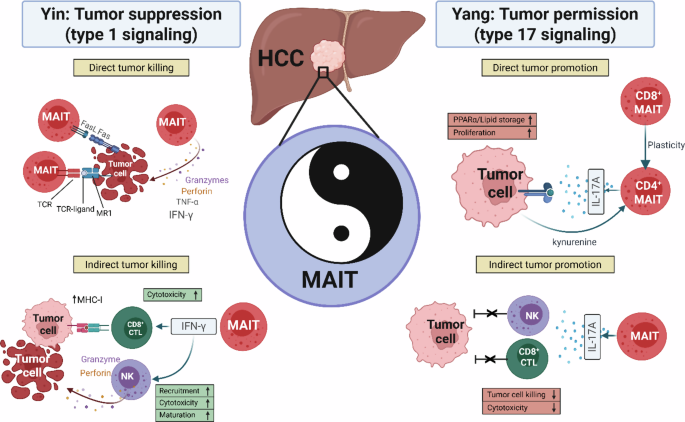
To identify potential molecular mechanisms driving MAIT cell plasticity in HCC, the authors compare transcriptomic data between CD8+ and CD4+ MAITs. Higher levels of AHR were detected on CD4+ MAIT cells than on other subsets, and this level is further elevated in HCC tumors. Stimulation with AHR agonists (FICZ or Kyn) increases glycolytic enzyme expression, glucose uptake, and IL-17A production. Seahorse metabolic flux analysis confirmed higher basal glycolysis and glycolytic capacity in CD4+ MAITs than in CD8+ or DN MAITs. At the molecular level, glycolysis promotes IL-17A production not by increasing the expression of the IL17A mRNA but through posttranscriptional regulation. Together, these findings describe a bidirectional metabolic feedback circuit between liver cancer cells and hepatic/tumor-infiltrating MAIT cells: tumor-derived Kyn → AHR activation in CD4+ MAITs → upregulation of glycolysis → IL-17A translation → PPARα/lipid-driven tumor proliferation.
The data by Fu et al. further support the notion that MAIT cells in HCC are neither simply protumor nor antitumor but are context-dependent effectors whose functional fate is shaped by the integration of multiple factors, such as TCR ligand engagement, the cytokine milieu, tumor-derived signals, the metabolic environment, and cellular interactions within the TME. In addition to the previous description of immunosuppressive MAITregs generated from conventional MAITs [10], the current data indicate that MAIT plasticity generates multiple subsets with tumor-promoting potential through distinct mechanisms. This study has important therapeutic implications: targeting immunosuppressive Kyn, e.g., through IDO1 inhibition, could decrease plasticity toward CD4+ MAIT-mediated tumor promotion, whereas anti-IL-17A strategies target downstream effector functions of CD4+ MAITs. Whether MAIT cell plasticity can also be reversed, e.g., by combining the use of MAIT TCR ligands and adjuvant treatment that promotes antitumoral TH1 responses (such as those involving IL-12 or IL-18), remains to be demonstrated. Overall, the results of the current study convincingly show that targeting the CD4+ MAIT–tumor metabolic circuit may represent a new method for improving the effectiveness of HCC immunotherapy.