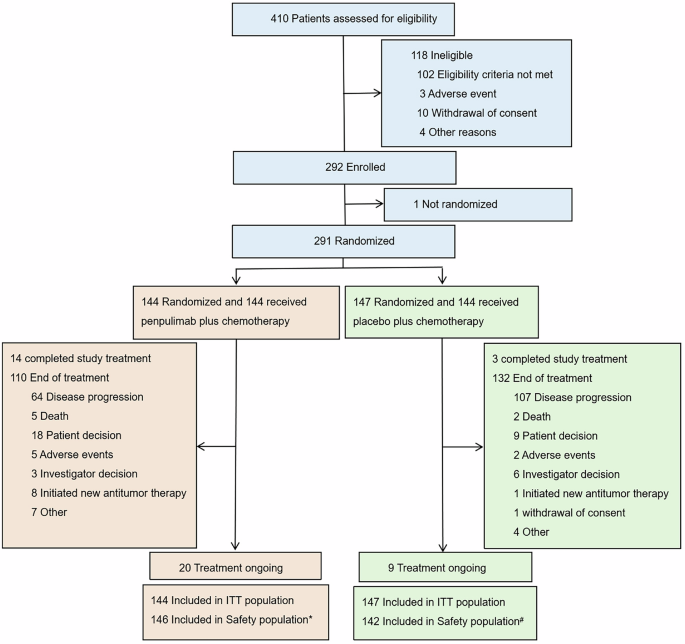
Study design and participants
This clinical trial (NCT04974398) was carried out following Good Clinical Practice standards and the principles outlined in the Declaration of Helsinki. The study protocol and all its modifications received approval from the independent ethics committees of the all sites involved in this study. The ethical registration number and the name of the approving committee or Institutional Review Board (IRB) are presented in supplementary Table 1. Additionally, all patients gave written informed consent prior to participating in the study.
In this research, patients with R/M NPC were enrolled from 36 sites in China and 10 sites outside of Asia. The details of all sites are shown in supplementary Table S1. Patients were eligible for enrollment as follows: aged 18–75 years; had histologically or cytologically confirmed NPC; had de novo metastatic NPC unsuitable for local management and so on. Detailed inclusion criteria and exclusion criteria were outlined in the study protocol.
Randomization and masking
Eligible patients were randomly allocated in a 1:1 ratio to take either penpulimab or placebo combined with chemotherapy. Permuted block randomization was carried out through an interactive web response system. Randomization was stratified based on disease stages (primary metastases versus recurrent), liver metastasis (present versus absent) and ECOG performance status (0 versus 1). The investigators, subjects, and other participants were blinded to treatment allocation unless unblinding was performed. To preserve the masking, the investigational drug and the matching placebo had identical packaging, labeling, appearance, and dosing regimen.
Procedures
Patients took intravenous penpulimab (200 mg on day 1) or matching placebo plus cisplatin (80 mg/m2 on day 1, Q3W) or carboplatin (AUC5 on day 1, Q3W) and gemcitabine (1000 mg/m2 on day 1 and day 8, Q3W) for a maximum of 6 cycles, followed by maintenance therapy with penpulimab 200 mg or placebo intravenously Q3W until no more clinical benefit was assessed by the investigator, intolerable toxic effects, completion of treatment, or meeting other criteria for discontinuation, whichever occurred first. In each arm, carboplatin was selected as a substitute for cisplatin based on the investigator’s discretion before randomization. Dose modification of penpulimab or placebo was not allowed during the treatment period. Dose modification and interruption of chemotherapy drugs for a maximum of 6 weeks were allowed. Subjects received penpulimab treatment for up to 24 months in this study. Subjects in the placebo group would have the option to transition to open-label penpulimab monotherapy after radiographic disease progression (confirmed by BICR) assessed by the investigators.
Tumor assessments based on imaging, including computed tomography (CT) or magnetic resonance imaging (MRI) of the nasopharynx, head, neck, chest, abdomen, and pelvis, were conducted every 6 weeks (±7 days) within 54 weeks after the first dose and every 9 weeks (±7 days) thereafter until radiographic progression or discontinuation of study treatment (whichever occurred later), initiation of new antitumor therapy, loss to follow-up, death, withdrawal of consent, or study discontinuation, whichever occurred first. If the symptoms of the subject worsened, an unscheduled imaging assessment was needed. If bone metastases were found at the screening and could not be seen on CT or MRI scans, or if clinically indicated, bone scans should be reexamined when a complete response was found or when progression in bone was suspected. If brain metastases were found at screening, the assessment was repeated at each subsequent tumor assessment. If no brain metastases were found at screening, an additional head CT scan or MRI might be performed during subsequent treatment if brain metastases were suspected.
Hematology assessments were completed no more than 3 days before dosing in each cycle (including day 1 and day 8, within a span of 2 days on day 8) and at the end of EOT visit. Blood biochemistry and urinalysis were conducted no more than 3 days before dosing in each cycle (day 1 only) and at the EOT visit. Thyroid function tests were conducted ≤3 days before dosing in each cycle and at the EOT visit except for cycle 1 day 1. The coagulation test was performed ≤3 days before dosing every 3 cycles and at the EOT visit and might be optionally performed by the investigator as clinically indicated. Adverse events (AEs) surveillance was performed continuously during treatment and for up to 30 days (90 days for SAE) after the last dose of study treatment (including chemotherapy) or the initiation of other antitumor therapy, whichever occurred first, immune-related AEs (irAEs) were collected until 90 days after the last dose of penpulimab (regardless of initiation of other antitumor therapies) and graded according to the National Cancer Institute Common Terminology Criteria for Adverse Events (NCI CTCAE), version 5.0. Survival was assessed every 3 months (±7 days) after drug discontinuation. PK samples and immunogenicity samples were collected within 60 min before the initiation of penpulimab in cycles 1, 2, 4, 8, and 14 and every 8 cycles thereafter. The EBV DNA level was analyzed in the central laboratory at cycles 1, 3, and 10 and at the EOT visit.
Outcomes
The primary endpoint of BICR-assessed PFS was measured from randomization until the earliest occurrence of disease progression per RECIST v1.1 or death from any cause. The key secondary endpoint was OS, defined as the time from the randomization date to death due to any cause. Other secondary endpoints included BICR-assessed objective response rate (ORR), defined as the proportion of subjects who achieved a best overall response (BOR) of CR or PR according to RECIST v1.1, duration of response (DoR), defined as the time from the first documented response (CR or PR) until disease progression based on RECIST v1.1 or death due to any cause, disease control rate (DCR), defined as the proportion of subjects who achieved the BOR of CR or PR or SD based on RECIST v1.1; investigator-assessed PFS, ORR, DoR and DCR based on RECIST v1.1; safety assessment, pharmacokinetic profile, immunogenicity, health-related quality of life (HRQoL), expression of PD-L1 in tumor samples, the relationship between PD-L1 expression and treatment effectiveness, the level of blood EBV at baseline and their changes following administration of the study drugs, as well as the relationship between blood EBV levels and treatment efficacy.
Statistical analysis
BICR-assessed PFS was tested with a two-sided alpha of 0.05. The trial was designed to enroll 278 patients and target 222 PFS events to achieve 89% power for detecting a hazard ratio (HR) of 0.65. The interim analysis for the BICR-assessed PFS was conducted when 189 PFS events were observed.
Since PFS reached statistical significance in this interim analysis, as planned in the statistical analysis plan, a descriptive analysis of OS, the key secondary endpoint, was conducted by spending a nominal α of 0.0001. No formal statistical testing or multiplicity adjustment was applied to other secondary endpoints. All efficacy analyses followed the ITT principle.
Adopting a stratified log-rank test to compare BICR-assessed PFS between two arms at the interim analysis with a significance threshold of 0.030. The Kaplan-Meier method was used to estimate the PFS, and HRs with corresponding 95% confidence interval (CI) were obtained from a stratified Cox proportional hazards model. The same statistical approach was applied to OS, investigator-assessed PFS, and BICR-assessed duration of response. Randomization stratification factors-disease stage (primary metastases vs. recurrent), liver metastasis status (present vs. absent), ECOG performance status (0 vs. 1), and chosen chemotherapy regimen (carboplatin vs. cisplatin) were included in all stratified models.
Prespecified subgroup analyses by age (under 50 or over 50 years), sex (male or female), ECOG performance-status score (0 or 1), liver metastases (yes or no), stage of disease at study entry (primary metastases vs. recurrent), investigator’s choice of chemotherapy regimens (carboplatin or cisplatin), PD-L1 expression level (TPS ≥ 50%) or negative (TPS < 50% or NA), EBV-DNA level (<500 copies/mL or ≥500 copies/mL), and region (Asian countries or non-Asian countries) were performed for BICR-assessed PFS. Unstratified Cox models provided HR estimates for each subgroup.
Health-related quality of life was assessed using EORTC QLQ-C30 and QLQ-H&N43 instruments. Change from baseline to week 30 was evaluated with a mixed-effects model for repeated measures, adjusting for stratification factors, baseline score, treatment, time, and treatment-by-time interaction. Time to deterioration in symptom scales was analyzed using methods consistent with the primary PFS analysis. For patients without confirmed worsening from baseline, the observation cutoff date is the date of the last observation. The EBV-DNA analysis population included all patients with baseline EBV-DNA results and at least one postbaseline result. Three binary variables (EBV DNA copy number decreased from baseline, EBV DNA copy number became undetectable after treatment, and EBV DNA rebounded after the initial reduction) were analyzed by the same method as for ORR. The analysis method for time from the lowest EBV copy number to rebound was the same as that for the primary endpoint PFS. Patients without rebound were reviewed as of the last sampling date.
The complete statistical analysis plan can be found in the supplementary material. All statistical analyses were performed using SAS version 9.4 (SAS Institute Inc.).