Lipid metabolic reprogramming, driven in part by AR-mediated transcriptional control of biosynthetic enzymes, is a recognized hallmark of PCa [19]. While lipidomic changes are well-characterized in primary tumors, the systemic lipidome in advanced stages, particularly mCRPC, remains poorly defined. Limited data on therapy-induced lipidomic shifts hinder understanding of AR-driven metabolic adaptations. Recent studies in both metastatic and localized PCa highlight the central role of lipid metabolism across disease stages [20, 21]. In this context, our study provides a comprehensive characterization of the systemic lipid landscape in patients with mCRPC, integrating longitudinal plasma lipidomic profiling during treatment with clinical outcomes, thereby reinforcing and extending this emerging line of evidence.
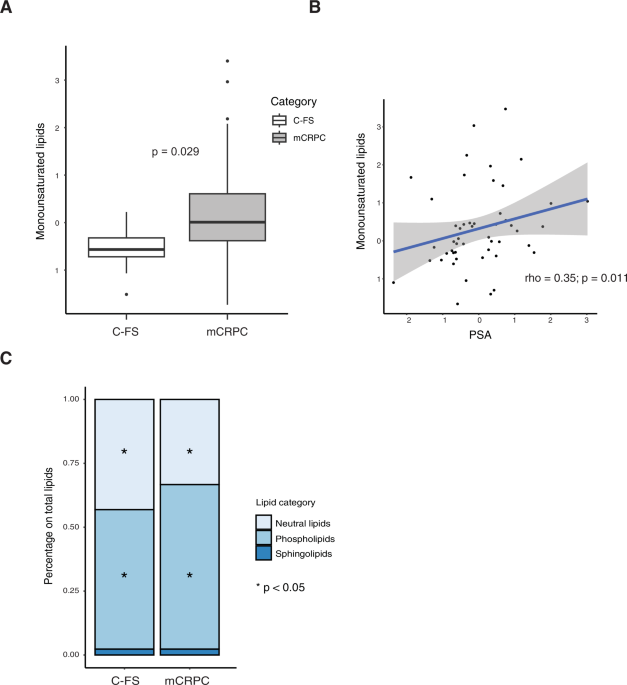
Profound alterations in the circulating lipidome of patients with mCRPC were observed, reflecting both tumor and therapy-driven remodeling. Compared to C-FS, mCRPC patients exhibit a distinct enrichment in monounsaturated and long-chain PL.
These changes, consistent with previous findings in primary PCa, suggest increased activity of AR-regulated enzymes such as fatty acid synthase (FASN), stearoyl-CoA desaturase (SCD), and elongases of very long-chain fatty acids (ELOVL) involved in denovo lipogenesis, membrane biogenesis, proliferation, and resistance to oxidative stress [22,23,24,25]. Moreover, circulating monounsaturated lipid levels also positively correlated with PSA concentrations, reinforcing their association with both tumor burden and clinical disease activity.
Lipid class analysis reveals a shift from energy storage toward membrane biosynthesis, with reduced levels of neutral lipids (e.g., triglycerides, cholesterol esters) and increased phospholipids. The relative enrichment of PE compared to PC within the phospholipid pool suggests a remodeling of membrane composition that prevents ER stress and the unfolded protein response in mCRPC, as well as enhance membrane fluidity. These changes, in part reversed by Enza treatment, could contribute to key malignant features such as increased proliferative capacity, invasive potential, and metastatic ability, ultimately supporting tumor cell adaptation and resistance to therapy. Elevated ceramides, increased in mCRPC, may modulate cellular response to treatment through their impact on apoptosis, cell-survival, and DNA repair regulation [26].
A major strength of our study lies in the longitudinal assessment of the systematic lipidomics during Enza treatment, combined with the clinical homogeneity of the cohort in terms of baseline characteristics and prior treatments, which enhances the reliability and interpretability of the findings.
Enza treatment was followed by a significant reduction of total lipids and monounsaturated fats, indicating suppressed lipid biosynthesis. It also induced coordinated remodeling across lipid classes, with decreased ceramides and major PLs, and increased SM—possibly reflecting a compensatory response to limit ceramide-induced cytotoxicity under AR inhibition [27]. In this context, the altered balance between SM and ceramides observed in our cohort may act as a sensor of cellular stress responses [27]. Moreover, androgen-dependent regulation of sphingolipid signaling has been previously demonstrated in prostate cancer models, supporting a functional interplay between androgen signaling and sphingolipid homeostasis [28, 29]
At radiographic progression, monounsaturated lipids began reverting toward pre- treatment levels, suggesting reactivation of resistance-associated metabolic programs
and highlighting the tumor’s metabolic plasticity. However, the limited sample size warrants further investigation.
The association between SL and clinical outcomes has been documented in previous studies, and these results corroborate these observations [26, 30,31,32,33,34]. Among the sphingolipid species, SM 18:1;18:1 emerged as a lipid with prognostic relevance. This sphingomyelin species is predominantly derived from the extracellular sphingomyelin pool via caveolin-1–mediated trafficking, linking its circulating levels to membrane dynamics and lipid exchange processes [35]. In line with these biological features, and consistent with the findings reported by Butler et al., SM 18:1;18:1 was associated with response to enzalutamide in patient-derived prostate cancer explants [12]. We independently validated that elevated circulating SM 18:1;18:1 during Enza treatment correlates with improved PFS and OS, even after adjusting for clinical covariates. This relationship may reflect a treatment-induced rebalancing of the ceramide–sphingomyelin axis, a pathway known to modulate cell fate decisions in cancer. Higher levels of SM 18:1;18:1 could mitigate the accumulation of bioactive ceramides that drive DNA damage, as we previously reported [15]. Furthermore higher SM 18:1;18:1 level may help stabilize membrane microdomains, supporting a sphingolipid environment that is less permissive to pro-survival and stress-response signaling.
While these results highlight distinct lipid alterations, these may arise both from the host, or the tumor itself. The clear response to Enza, however, strongly suggest a tumor origin reflected in serum lipids. This has been clearly demonstrated in mouse models of PCa [13, 36]. Thus, these findings support plasma lipidomics as an indirect surrogate of tumor metabolic state. Over and above their biomarker value for monitoring disease progression, the circulating lipidome may also inform therapeutic strategies, particularly in patients under AR-targeted therapy. In addition, ongoing trials targeting lipid metabolism further underscore the therapeutic relevance of this pathway in PCa (NCT05743621) [16].
Although many patients in the mCRPC cohort had progressed on ADT monotherapy—no longer recommended as first-line treatment for mCRPC—the lipidomic changes observed following androgen deprivation likely reflect fundamental androgen-regulated metabolic alterations that are maintained under contemporary, intensified therapeutic regimens. Accordingly, the lipid signatures identified in this study may capture intrinsic aspects of advanced PCa biology rather than effects strictly dependent on a specific treatment sequence.
While the study provides novel insights on the dysregulation of lipid metabolism and its potential utility in the clinical management of advanced PCa, some considerations remain. The control cohort was not stratified by clinical parameters, introducing potential variability. The small metastatic cohort size, especially the progressive subgroup, may limit generalizability despite cohort homogeneity. Additionally, unrecorded dietary habits and medications could have influenced lipidomic profiles.