Prostate cancer exceeds 1.4 million new cases annually worldwide, yet access to high-quality surgical treatment is uneven [2]. Over the past two decades, robotic surgery has become the gold standard for localized prostate cancer [3], but in many low- and middle-income settings, the scarcity of trained surgeons limits the impact of deployed systems. Telesurgery aims to bridge this expertise gap by enabling remote operation and expanding access to expert-level care [13,14,15].
Despite early successful initiatives [1, 4], the adoption of telesurgery in urology has not progressed as rapidly as robotic surgery overall. Its broader implementation has been hindered by unresolved regulatory, ethical, and logistical challenges that remain particularly relevant in cross-border or resource-limited contexts [16]. In addition to regulatory, ethical, and logistical barriers, cost remains one of the most significant challenges to the wider adoption of telesurgery [17].
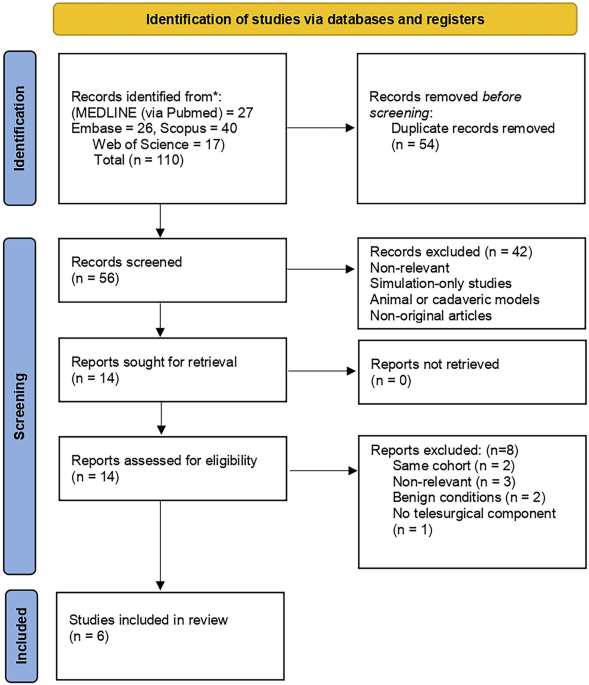
This systematic review aims to comprehensively assess the current evidence on telesurgery in the management of prostate cancer. We highlight its feasibility and safety across various procedural platforms including RARP and HIFU while also identifying key limitations, regulatory constraints, and future considerations for wider clinical adoption.
Early telesurgical research in porcine models shaped the clinical development of remote prostate surgery. Marescaux et al.’s landmark transatlantic operation using the ZEUS system demonstrated that complex procedures can be performed safely across long distances [1]. Although not urologic, this pioneering effort provided a compelling technological proof of the telesurgery concept. Building on this foundation, Sterbis et al. performed the first urologic telesurgical procedures in porcine models using the da Vinci system [4], highlighting the field’s sensitivity to network performance. The central determinant of safe telesurgical performance is end-to-end latency, defined as the interval between a console command and the instrument’s response. Functional control is generally maintained when latency remains below the 200 ms threshold; higher delays can disrupt psychomotor coordination and visual feedback [18]. Consistent with this threshold, Sterbis et al. demonstrated in porcine models that delays approaching 900 ms caused lagging instrument response and suboptimal console visualization, underscoring the risks associated with high-latency environments [4].
With modern networks, feasibility improves. Patel et al. demonstrated that complex urologic procedures can be performed across the Pacific in porcine models using hybrid 5 G and fiber-optic links; consistently low latency enabled uninterrupted surgical flow without perceptible delay or control issues [19]. Collectively, these preclinical experiences clarified the importance of latency thresholds, infrastructure demands, and workflow design, laying a foundation for translating telesurgical RARP into human clinical practice.
Building on the experimental groundwork, early human telesurgical prostate procedures have yielded encouraging results. Across clinical telesurgical cases, average latency remained below the 200 ms threshold. Moschovas et al [8]. reported a median round-trip latency of 22 ms, whereas Aldousari et al. [6], Moschovas et al. [7], and Olivares et al [10]. reported mean round-trip latencies of 181 ms, 6–8 ms, and 115 ms, respectively, without audiovisual disruption or instrument lag. Brief spikes above 200 ms occurred in some reports [5, 6] but did not interrupt surgery, likely because the mean latency stayed within target range. These observations support that while peaks can occur, average latency may be a more meaningful indicator of telesurgical performance than isolated spikes [20]. Taken together, contemporary infrastructures, including 5 G and SD-WAN, increasingly support real-time telesurgical workflows across diverse distances.
Ensuring uncompromised operative outcomes is critical when telesurgery is used to deliver care in underserved regions where access to high-volume expertise is limited. In our review, clinical results from telesurgical prostate procedures were within expected ranges for contemporary cohorts, supporting feasibility and a favorable safety profile. Across reported telesurgical RARP cases [5,6,7,8,9], key perioperative parameters, including operative time, estimated blood loss, intraoperative and postoperative complication rates, length of stay, and positive margin status, were within accepted benchmarks and consistent with contemporary robotic cohorts. Where reported, procedures were completed without conversion. The single telesurgical HIFU case also showed a favorable intraoperative course and rapid recovery, with discharge at 4 h and no compromise of clinical safety or oncologic adequacy [10]. Collectively, these findings suggest that, with appropriate infrastructure and team configuration, telesurgery can reproduce expected perioperative standards while extending expert-level care across distance.
Among the reported cases, the triple-console telesurgical RARP performed by Rogers et al. represents a notable advancement in collaborative remote surgery [9]. By enabling three surgeons across different locations to participate in real time, the setup demonstrated not only technical feasibility but also educational potential. This configuration allowed for dynamic intraoperative collaboration and may support mentorship during live procedures, aligning with previous findings that multi-console systems enhance surgical training and safety [21, 22]. As telesurgery evolves, such architectures could play a critical role in promoting global expertise sharing and team-based surgical care [9].
In parallel with the evolution of surgical techniques, the robotic platforms utilized in telesurgical urologic surgery have also diversified in terms of design philosophy, console configuration, and collaborative capacity [5,6,7,8,9]. The Edge MP1000 (Shenzhen Edge Medical Co., Ltd., Shenzhen, China) features a closed-console setup and integrated communication architecture, supporting efficient single-surgeon telesurgical prostatectomy without visual or control delays [8]. The KD-SR-01 system offers an open-console design and supports triple-console functionality, allowing multiple surgeons to participate remotely in real time. Although initial setup and console times may be slightly longer, the system maintains clinical safety and offers a favorable learning curve [23]. In comparison, the TRSS platform (Shanghai MicroPort MedBot (Group) Co., Ltd., Shanghai, China) supports dual-console use and has demonstrated success in ultra-long-distance telesurgery, highlighting its potential for distributed collaboration and training scenarios [24]. Beyond its ultra-long-distance application, the same TRSS (Toumai) platform has also been used in a same-city configuration in Luanda, Angola, achieving single-digit latency over dedicated fiber and uneventful perioperative courses [7]. A broader overview of representative telesurgery systems and the level of supporting evidence is provided in Table 4. These considerations form an important backdrop for understanding the telesurgical cases included in this review.
Table 4 Representative robotic and energy-based systems relevant to telesurgery.
Reported experience underscores that communication instability is a distinctive intraoperative risk in telesurgery. A key limitation of telesurgical RARP is the dependence on an on-site team for time-critical management of intraoperative complications. The workflow also introduces communication-related failure risks. If connectivity is lost or network conditions compromise safe teleoperation, predefined protocols should enable rapid bedside takeover and, when necessary, conversion to open surgery or discontinuation of the remote procedure [16]. In reported clinical implementations, this risk is mitigated through extensive pre-procedure system testing (including repeated phantom-model trials) and redundancy measures such as backup connectivity and a separate audiovisual feed [10]. By contrast, HIFU offers a practical advantage by eliminating tissue excision and thereby removing the risk of emergent conversion; if technical or procedural issues arise, the intervention can be safely aborted without another surgeon assuming control [25,26,27]. As an image-guided and minimally invasive modality, HIFU minimizes intraoperative bleeding and streamlines logistics, making it particularly well suited for remote prostate interventions [28, 29]. In the transcontinental case by Olivares et al. using the robotic Focal One system, focal HIFU for localized prostate cancer was delivered over ≈11,412 km without complications and with discharge at 4 h, illustrating procedural efficiency without compromising safety and highlighting potential scalability for focal telesurgical therapies [10, 29].
Despite these promising developments and successes, several issues must still be addressed before telesurgery can be widely adopted into routine clinical practice. The ethical use of telesurgical systems depends not only on technological capabilities but also on clear institutional responsibilities, open communication, and appropriate regulatory oversight. Patel et al. highlight that beyond secure network infrastructure, it is essential to clearly define the roles of all involved parties, including remote and on-site surgeons, technical teams, and device manufacturers, at all stages of the procedure: before, during, and after [30]. This is especially important when surgeries take place across different regions or countries, where licensure requirements, liability rules, and data protection standards may differ. Ensuring patient safety in such contexts requires mutual understanding around informed consent, control transfer protocols, and emergency planning [31, 32].
Accordingly, accountability for intraoperative adverse events should be prospectively allocated between the remote console surgeon and the on-site team, and documented within institutional credentialing and telesurgery-specific consent materials. In emergency scenarios, the authority to initiate immediate takeover, undocking, or conversion should be explicitly defined in advance, with the bedside team empowered to act without delay when patient safety thresholds are met. Such role delineation is especially critical in cross-border telesurgery, where professional liability, jurisdiction, and malpractice coverage may not align across participating sites [30,31,32]. However, the legal landscape for telesurgery remains underdeveloped. There is currently no unified international framework governing cross-border telesurgical practice, and informed-consent documents often insufficiently address telesurgery-specific issues such as the operator’s physical absence, potential technical failures, and data-security risks [33, 34]. As noted by Aldousari et al., coordinated action among regulators, legal experts, and clinical societies is essential to close these gaps and enable safe, scalable, and legally sound implementation [6].
Cybersecurity is a prerequisite for scalable telesurgery because real-time patient data and robotic control signals may be vulnerable to unauthorized access or tampering. Mitigation measures include operating over dedicated or logically isolated networks, end-to-end encryption (for example, IPsec), strict access control, and continuous monitoring with intrusion detection or prevention systems, alongside predefined fail-safe pathways for immediate on-site takeover if security or connectivity thresholds are breached [35]. Experimental integration of intrusion prevention devices and IPsec in a telesurgical setup has been shown to detect simulated attacks without compromising operability or image quality, while adding only minimal delay and without a meaningful increase in communication error rates [35]. Emerging approaches also propose privacy-preserving intrusion detection using federated learning, although real-world clinical validation remains limited [36].
Cost continues to represent another dominant barrier to the broader utilization of telesurgery. Although robotic surgical systems are increasingly being deployed globally through public and private investments, their integration into telesurgical programs requires substantial infrastructure [30]. This includes high-speed broadband, telecommunication redundancy, robotic platforms, and latency optimization technologies. These requirements are especially burdensome in low- and middle-income countries, where digital infrastructure and surgical workforce capacity remain limited [17, 19, 37]. Bart et al. further emphasize that concerns regarding financial sustainability and inadequate funding frameworks continue to hinder implementation in these regions [38].
Nevertheless, emerging literature suggests that telesurgery may yield long-term economic value by reducing travel for patients and providers, improving resource allocation, and centralizing training, which together could offset upfront costs [38, 39]. Continued advances in robotics, networks, and immersive tools may enable safe telesurgery outside tertiary centers, including in remote settings [8]. Realization will require coordinated efforts by industry, health systems, and regulators to establish scalable, ethical, and legally robust frameworks [6]. Ultimately, adoption depends on technical readiness, licensure harmonization, cost-effectiveness, and public trust, enabling more equitable, expert-driven surgical care [9].
This review has several limitations that should be acknowledged. First, the included evidence is derived from a small number of clinical cases, which limits the generalizability of the findings and precludes robust statistical analysis. Due to the narrative nature of this review, outcomes were synthesized qualitatively rather than through pooled or comparative data. Moreover, none of the included telesurgical prostatectomy studies were designed as direct head-to-head comparisons with conventional in-person procedures, making it difficult to assess the true magnitude of any potential differences in clinical efficacy or safety. Finally, long-term oncologic and functional outcomes remain unreported in the existing literature, thereby restricting the ability to evaluate the durability of telesurgical interventions in prostate cancer management. Future prospective studies with larger cohorts, standardized outcome measures, and longer follow-up are essential to validate these early observations and support broader implementation.