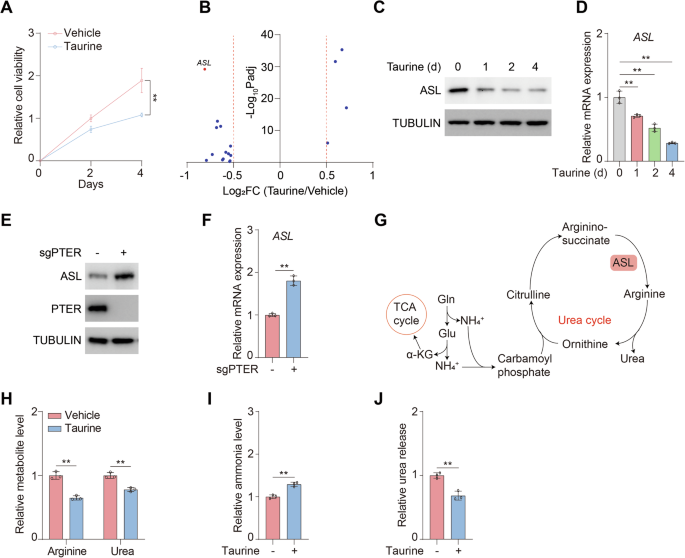
Taurine negatively regulates urea cycle
Since the abundance of taurine in complete media (Fig. S1A) was far below the physiological concentration [8], to explore the role of taurine on cell proliferation, we treated hepatocellular carcinoma cancer cells MHCC97H and HepG2 with 100 μM taurine for consecutive days and found that taurine was sufficient to restrain cell proliferation (Figs. 1A and S1B). To reveal the molecular mechanism in depth, we conducted RNAseq analysis and found that ASL, the key enzyme of urea cycle, was strikingly decreased upon taurine treatment (Fig. 1B). To validate the results, we found that the protein (Figs. 1C and S1C) and mRNA (Figs. 1D and S1D) levels of ASL was gradually declined under taurine treatment.
Fig. 1: Taurine negatively regulates urea cycle.
A Cell viability of MHCC97H cells treated with or without 100 μM taurine for the indicated days were analyzed. B RNAseq demonstrated that the expression of ASL was significantly changed among various metabolic enzymes in MHCC97H cells treated with or without 100 μM taurine for four days. Volcano plot was displayed with log2FC > 0.5 or <-0.5. C, D MHCC97H cells were treated with or without 100 μM taurine for the indicated days. Immunoblotting analysis was performed using the indicated antibodies (C). mRNA levels of ASL were analyzed (D). E, F MHCC97H cells were transfected with or without sgRNA targeting PTER (sgPTER). Immunoblotting analysis was performed using the indicated antibodies (E). mRNA levels of ASL were analyzed (F). G Schematic of ASL-mediated urea cycle. H-J MHCC97H cells were treated with or without 100 μM taurine for four days. Arginine and urea levels (H) ammonia levels (I) and urea release (J) were analyzed. Data are presented as mean ± SD, n = 3 independent repeats. Unpaired, two-tailed t test; **P < 0.01.
PTER has been demonstrated as N-acetyltaurine hydrolase to generate taurine and regulate obesity [27]. We therefore established PTER knockout (sgPTER) HCC cell lines (Figs. 1E and S1E) and found that loss of PTER enhanced expression of ASL (Figs. 1E, F and S1E, F). Since ASL is required for maintaining homeostasis of urea cycle, which is the major route for ammonia disposal and production of arginine and urea (Fig. 1G), we examined the cellular metabolites and found that taurine largely reduced cellular arginine and urea levels (Figs. 1H and S1G). Consistently, taurine prominently enhanced ammonia accumulation (Figs. 1I and S1H). As a result, urea released was disrupted by taurine (Figs. 1J and S1I). Taken together, taurine represses urea cycle and expression of ASL.
Metabolic impacts of taurine are linked to expression of ASL
To verify whether ASL mediated metabolic impacts of taurine, we conducted ASL knockdown (shASL) HCC cell lines (Figs. 2A and S2A). The results showed that knockdown of ASL compromised taurine-induced reduction of arginine and urea levels (Figs. 2B and S2B). Likewise, the ammonia levels (Figs. 2C and S2C) and urea release (Figs. 2D and S2D) were accordingly orchestrated by taurine in an ASL-dependent manner. Consequently, ASL was involved in taurine-mediated inhibition of cell proliferation. Compared with control group, loss of ASL presented mild reduction of cell viability upon taurine treatment (Figs. 2E and S2E).
Fig. 2: Metabolic impacts of taurine are linked to expression of ASL.
A MHCC97H cells were transfected with or without shRNA targeting ASL (shASL). Immunoblotting analysis was performed using the indicated antibodies. B-D shCtrl and shASL MHCC97H cells were treated with or without 100 μM taurine for four days. Arginine and urea levels (B) ammonia levels (C) and urea release (D) were analyzed. E Cell viability of shCtrl and shASL MHCC97H cells treated with or without 100 μM taurine for indicated days were analyzed. F MHCC97H cells were stably overexpressed with or without ASL (ASL OE) and treated with or without 100 μM taurine for two days. Immunoblotting analysis was performed using the indicated antibodies. G-I Control and ASL OE MHCC97H cells were treated with or without 100 μM taurine for two days. Arginine and urea levels (G) ammonia levels (H) and urea release (I) were analyzed. J Cell viability of control and ASL OE MHCC97H cells treated with or without 100 μM taurine for indicated days were analyzed. Data are presented as mean ± SD, n = 3 independent repeats. Unpaired, two-tailed t test; *P < 0.05; **P < 0.01. NS, not significant.
Next, we generated HCC cell lines stably expressing ASL, which were identified resistant to taurine-induced suppression during transient treatment (Figs. 2F and S2F). Overexpression of ASL significantly facilitated arginine and urea production in control group (Figs. 2G and S2G). Moreover, ASL was capable of sustaining urea cycle in response to taurine (Figs. 2G and S2G). Besides, overexpression of ASL maintained ammonia consumption regardless of taurine (Figs. 2H and S2H). Accordingly, urea release was enhanced by ASL, whereas taurine failed to impair urea release under forced expression of ASL (Figs. 2I and S2I). In regard to cell viability, overexpression of ASL accelerated cell proliferation and resisted the suppressive effect of taurine compared with the control group (Figs. 2J and S2J). ASL functioned as the critical enzyme in urea cycle to catalyze arginine and urea generation (Fig. 1G) and enabled tumor growth [22, 28]. Moreover, a previous study has demonstrated that sole knockdown of ASL is sufficient to disrupt urea cycle and proliferation of HCC cells [22]. These findings indicated that manipulation of ASL expression enables the orchestration of urea cycle. Considering that ASL has been revealed as a crucial target of taurine and was responsible for the suppressive effect on urea cycle caused by taurine, exogenous forced expression of ASL was capable of maintaining urea cycle homeostasis and cell viability and presented resistant to taurine treatment. Collectively, ASL plays a pivotal role in mediating the metabolic impacts of taurine.
Taurine represses expression of ASL via FOS
To decipher the mechanism of taurine-triggered suppression of ASL transcription, we predicted the putative transcription factor of ASL and found that ASL promoter harbored two consensus binding sites of FOS:JUN complex (Fig. 3A). Interestingly, RNAseq revealed that taurine remarkably restrained expression of FOS, which provoked us to explore whether FOS:JUN complex could initiate transcription of ASL. In line with the RNAseq results, the expression of FOS was substantially reduced by taurine (Figs. 3B, C and S3A, B). Furthermore, luciferase reported assay assured FOS could associate with ASL promoter, while mutation of the putative sites entirely blocked the interaction (Fig. 3D).
Fig. 3: Taurine represses expression of ASL via FOS.
A Putative FOS:JUN binding consensus sequences in promoter of ASL (bottom) were compared with motif predicted using JASPAR database (top). B, C MHCC97H cells were treated with or without 100 μM taurine for the indicated days. Immunoblotting analysis was performed using the indicated antibodies (B). mRNA levels of FOS were analyzed (C). D Plasmid containing either WT or mutant promoter sequence of ASL was co-transfected with FOS into 293T cells. Dual luciferase reporter assay was performed. E, F MHCC97H cells were transfected with or without sgRNA targeting FOS (sgFOS) and treated with or without 100 μM taurine for four days. Immunoblotting analysis was performed using the indicated antibodies (E). mRNA levels of ASL were analyzed (F). G ChIP assay was performed in sgCtrl and sgFOS MHCC97H cells using antibodies against c-JUN. DNA enrichment was examined by quantitative real-time PCR. The y axis shows the value normalized to input. H-J sgCtrl and sgFOS MHCC97H cells were treated with or without 100 μM taurine for four days. Arginine and urea levels (H) ammonia levels (I) and urea release (J) were analyzed. K Cell viability of sgCtrl and sgFOS MHCC97H cells treated with or without 100 μM taurine for indicated days were analyzed. Data are presented as mean ± SD, n = 3 independent repeats. Unpaired, two-tailed t test; *P < 0.05; **P < 0.01. NS, not significant.
Next, FOS knockout (sgFOS) HCC cell lines were carried out (Figs. 3E and S3C). As a result, depletion of FOS impaired expression of ASL, while compare with control group (sgCtrl), taurine exerted minor effect on expression of ASL in sgFOS group (Figs. 3E, F and S3C, D). Moreover, chromatin immunoprecipitation (ChIP) analysis indicated that c-JUN was abundantly enriched on the promoter of ASL, which was compromised upon loss of FOS (Figs. 3G and S3E). In regard to the metabolic effects, deficiency of FOS notably impaired arginine and urea production, which phenocopied the effect of taurine treatment (Figs. 3H and S3F). Moreover, loss of FOS disrupted the reduction of arginine and urea caused by taurine (Figs. 3H and S3F). Consistently, taurine coordinated ammonia abundance (Figs. 3I and S3G) and urea release (Figs. 3J and S3H) in a FOS-dependent manner. Consequently, deletion of FOS significantly suppressed cell viability, while taurine exerted a mildly suppressive effect on proliferation capacity of sgFOS cells (Figs. 3K and S3I). Together, FOS is critical for taurine-mediated suppression of urea cycle.
FOS-ASL axis contributes to metabolic effects of taurine
To validate the role of FOS-ASL axis in mediating the metabolic impacts of taurine, we tried to introduce ectopic ASL into FOS knockout cells (Figs. 4A and S4A). Overexpression of ASL considerably rescued arginine (Figs. 4B and S4B) and urea (Figs. 4C and S4C) production under either taurine treatment or deletion of FOS. Moreover, ASL could entirely support urea cycle in FOS knockout cells with taurine treatment (Figs. 4B, C and S4B, C), implying the FOS-ASL axis was vital for taurine in regulation of urea cycle. Likewise, forced expression of ASL reversed the ammonia accumulation (Figs. 4D and S4D) and urea release (Figs. 4E and S4E) caused by taurine or FOS knockout. Ultimately, overexpression of ASL fueled cell proliferation regardless of taurine treatment or loss of FOS (Figs. 4F and S4F). These findings indicated that FOS-ASL axis contributes to metabolic effects of taurine.
Fig. 4: FOS-ASL axis contributes to metabolic effects of taurine.
A-F MHCC97H cells were transfected with or without sgRNA targeting FOS (sgFOS), overexpressed with or without ASL (ASL OE) and treated with or without 100 μM taurine for two days. Immunoblotting analysis was performed using the indicated antibodies (A). Arginine (B), urea (C), ammonia levels (D), urea release (E), and cell viability (F) were analyzed. Data are presented as mean ± SD, n = 3 independent repeats. Unpaired, two-tailed t test; **P < 0.01. NS, not significant.
Taurine enhances the efficacy of glutaminolysis blockade
Subsequently, the physiological consequences of taurine-mediated metabolic impacts were explored via xenograft analysis. The results showed that taurine solely was sufficient to suppress tumor growth (Fig. 5A, B). Compared with control group, taurine failed to exert an additional suppressive effect on tumors with knockdown of ASL (Fig. 5A, B), suggesting ASL was the main metabolic target in orchestrating tumor growth.
Fig. 5: Taurine enhances the efficacy of glutaminolysis blockade.
A, B shCtrl and shASL MHCC97H cells were subcutaneously injected into athymic nude mice administrated with or without taurine. Representative tumor xenografts (A). Mice were sacrificed in week 4 and weight were calculated (B). n = 6 independent animals. C Schematic of GLS1-mediated glutaminolysis, which can be blocked by its inhibitor CB839. D MHCC97H cells were treated with or without 500 nM CB839 or 100 μM taurine for four days. Arginine and urea levels were analyzed. E Cell viability of MHCC97H cells treated with or without 500 nM CB839 or 100 μM taurine for indicated days were analyzed. F, G MHCC97H cells were subcutaneously injected into athymic nude mice administrated with or without CB839 or taurine. Representative tumor xenografts (F). Mice were sacrificed in week 4 and weight were calculated (G). n = 6 independent animals. H Schematic of synergistic blockade of glutaminolysis and urea cycle to suppress tumor growth. Data are presented as mean ± SD, n = 3 independent repeats. Unpaired, two-tailed t test; *P < 0.05; **P < 0.01. NS, not significant.
Since glutaminolysis catalyzed by GLS1 fuels TCA cycle, urea cycle and other physiological provesses via producing α-KG and ammonia (Fig. 5C), inhibition of glutaminolysis has been considered as a promising route for cancer therapy [29]. Nevertheless, due to the limited efficacy of GLS1 inhibitor, there is an urgent demand to improve the efficacy. Recently, preclinical studies have revealed that radiation therapy and drugs targeting redox stress have been utilized to synergize with GLS1 inhibitor for cancer therapy [30, 31]. Since glutamine is the major source of ammonia to fuel urea cycle [32], we proposed that dual blockade of glutaminolysis and urea cycle may be a promising route for cancer therapy. CB839 (Fig. 5C), the inhibitor of GLS1 which catalyzes the first step of glutaminolysis [33], was employed to verify the hypothesis. Firstly, we found that either taurine or CB839 exerted no effect on GLS1 expression (Fig. S5A, B) and its endogenous activity as indicated by cellular glutamate/glutamine ratio (Glu/Gln) (Fig. S5C) or ASL expression (Fig. S5D, E), respectively. Interestingly, CB839 could notably dampen urea cycle capacity, which was strengthened by taurine (Fig. 5D). In line with this, CB839 plus taurine substantially impaired cell viability compared with CB839 solely (Fig. 5E). In regard to in vivo validation using xenograft model, compared with CB839 group, synergistic combination of CB839 and taurine exhibited a prominent inhibitory effect on tumor growth (Fig. 5F, G). Together, these results demonstrated that taurine could enhance the efficacy of glutaminolysis blockade to repress tumor growth (Fig. 5H).